Translate this page into:
Imaging appearance of pre-osseous and osseous phase of fibrodysplasia ossificans progressiva: A rare entity

*Corresponding author: Pankaj Sharma, Department of Diagnostic and Interventional Radiology, All India Institute of Medical Sciences Rishikesh, Rishikesh, Uttarakhand, India. pankajrad7477@yahoo.com
-
Received: ,
Accepted: ,
How to cite this article: Bhardwaj A, Sharma P. Imaging appearance of pre-osseous and osseous phase of fibrodysplasia ossificans progressiva: A rare entity. Indian J Musculoskelet Radiol. doi: 10.25259/IJMSR_25_2024
Abstract
Fibrodysplasia ossificans progressiva (FOP) is an infrequent autosomal dominant disorder, characterized by progressive heterotopic ossification of soft tissues including skeletal muscles, tendons, ligaments, fascia, and aponeuroses. Clinically and radiologically, it may be misdiagnosed a soft-tissue neoplasm or inflammatory/infective myositis. The knowledge and awareness of early imaging appearances, soft tissue, and skeletal deformities can lead to early diagnosis before ossification starts to occur, leading to dedicated treatment and prevention of unnecessary biopsies/iatrogenic trauma. We describe a rare case of clinically and radiologically diagnosed fibrodysplasia ossificans progressiva (FDOP) in a 5-year-old girl. This report highlights the radiological findings and the implications of this disorder.
Keywords
Congenital
Autosomal dominant
Heterotopic ossification
Muscle edema
Soft tissue swellings
Pre-osseous phase
Osseous phase
INTRODUCTION
Fibrodysplasia ossificans progressiva (FOP) is an infrequent autosomal dominant disorder, characterized by typical skeletal malformations of progressive heterotopic ossification of soft tissues including skeletal muscles, tendons, ligaments, fascia, and aponeuroses.[1] Magnetic resonance imaging (MRI) and ultrasound may aid in early diagnosis, during episodes of flare ups when patient presents with soft tissue swellings before ossification is visible on radiographs.
CASE REPORT
A 5-year-old girl presented with complaints of painful swelling in the left shoulder region for the past 15–20 days which gradually developed after an episode of fall while playing [Figure 1a]. She subsequently developed similar swellings in the left scapular region, left chest wall, right shoulder, and right side of the neck. The patient was afebrile. On examination, the swellings were tender with associated local rise in temperature and erythema. The lower limbs showed no visible swelling or wasting but with shortened great toes [Figure 1b]. The patient carried an MRI report done at an outside center which was reported as myositis with cellulitis. Images were not available for review. Patient’s blood work up was sent and intravenous antibiotics were started with a provisional diagnosis of infective myositis. The blood counts, C- reactive protein, erythrocyte sedimentation rate, and creatine phosphokinase levels were within normal range.
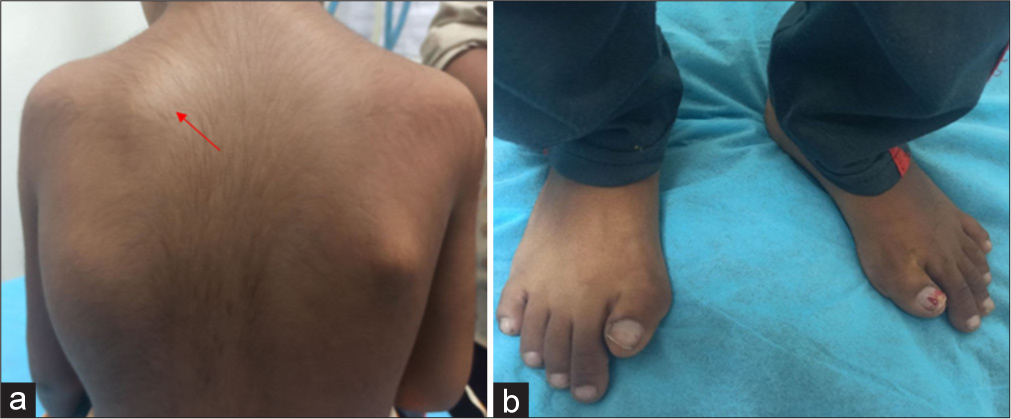
- Clinical pictures of patient with fibrodysplasia ossificans progressive.(a) Soft tissue mass in the left scapular region.(red arrow). (b) Bilateral shortened great toe with hallux valgus deformity.
With no improvement in the clinical symptoms, the patient was referred to the Radiology department for an MRI of both shoulders and chest wall. The MRI [Figure 2] revealed hyper intense edematous changes in the left scapular region extending along the left chest wall, involving the subcutaneous plane as well as underlying muscle, extending along the subcutaneous plane and muscular plane of the chest wall on the left side. The patient was called subsequently for a contrast scan. It is noteworthy that a prompt increase in size of the mass was evident on MRI, within a span of 6 days between the time point of the non-contrast and contrast study. Extensive edema and enhancement was also seen along the muscle fibers and intervening fascial planes of bilateral rotator cuff muscles (right side being involved more than left), upper arms, and pectoralis major muscles [Figure 3]. A non-enhancing focal T1/T2 hypointense area was seen in the left coracobrachialis, for which ultrasound correlation was performed that showed coarse echogenic areas with posterior acoustic shadowing suggesting coarse calcification [Figures 4 and 5]. The rest of the involved muscles appeared edematous. Similar calcifications were also seen in the infraclavicular region along the pectoralis muscles. Subsequently, a skeletal survey [Figure 6] was also performed which revealed bilateral shortened first metatarsals, mono-phalangeal great toe with hallux valgus deformity, bilateral shortened second and fifth digits with reduced length of the respective proximal phalanges. In addition, there was heterotopic ossification bridging across the left first metacarpophalangeal joint and along the soft tissues of both shoulders, infraclavicular region, and paraspinal region in the neck.
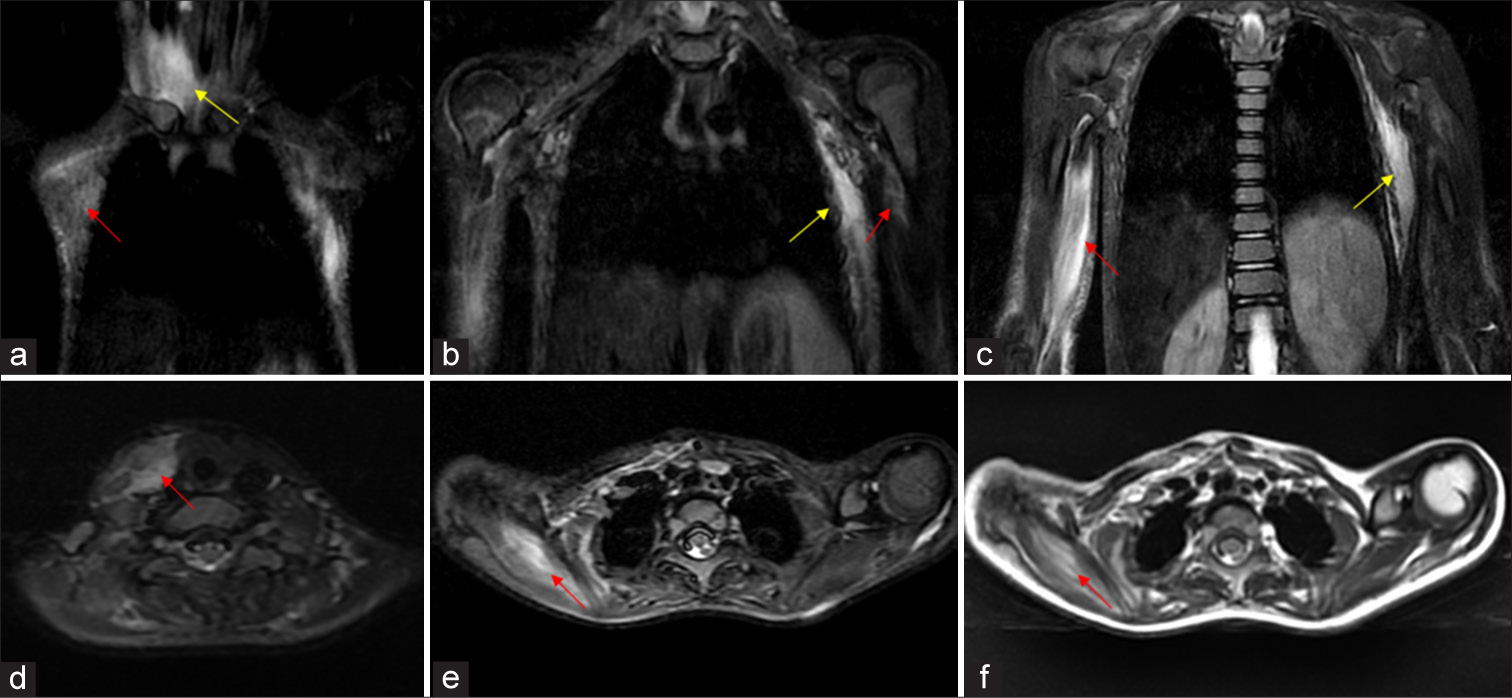
- MRI of bilateral shoulders and chest wall showing pre-osseous soft tissue changes in 5 year old child with FOP. (a-c) Serial coronal STIR images shows hyperintense signal (red arrow) along the fibres of right sternocleidomastoid (a, yellow arrow), left coracobrachialis (b red arrow), right coracobrachialis and biceps and overlying subcutaneous fat (c red arrow), subcutis and intercoastal muscles of left side of chest wall (b, c yellow arrows). (d-e) Serial axial STIR images shows edema (red arrow) along the right sided strap muscles and sternocleidomastoid, bilateral supraspinatus (Right much more than left). (f) Axial T2 image shows hyperintense signal (red arrow) along the fibres of right supraspinatus, which also appears bulky due to the edema.
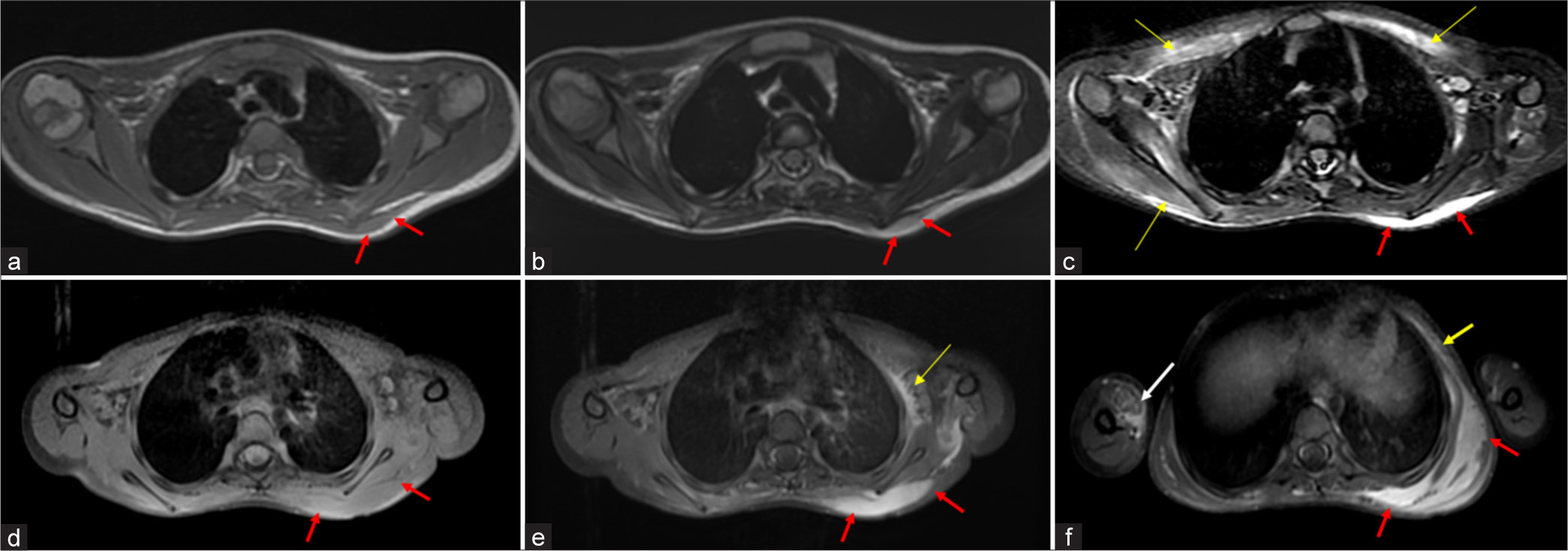
- MRI of bilateral shoulders and chest wall showing pre-osseous soft tissue lesion in 5 year old child with FOP. (a) Axial T1WI shows a soft tissue mass (red arrows) in the left scapular region in the subcutaneous plane which is iso-intense to the muscle. (b) Axial T2WI and (c) corresponding axial STIR image shows the mass (red arrows) to have a hyperintense signal. STIR hyperintense edema is also seen in the pectoralis major muscles and right rotator cuff muscles (yellow arrows). (d) Axial pre-contrast T1 fat suppressed image and (e-f) post contrast images shows intense enhancement of the soft tissue lesion, which is seen to extend along the trapezius muscle, left intercoastal muscles(yellow arrow) and overlying subcutis of the chest wall. (d-f) Note that the contrast study were performed 6 days after the initial base sequences(a-c) and shows a significant increase in size. Enhancement was also seen within the involved right arm muscles (f: white arrow) and rest of the involved muscles which were edematous (not shown here). Pre-osseous mass scapular region 6-7-21, Scapular mass contrast 12-7-21.
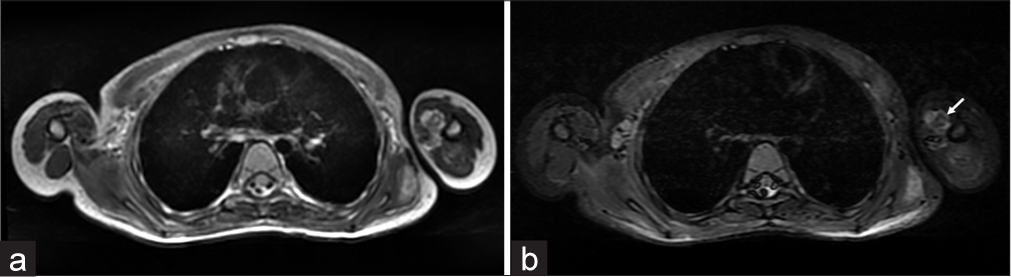
- MRI showing ossific foci left arm muscles. (a) Axial T2WI and (b) STIR image showing central hypointense foci(white arrows) within the hyperintensity in the left coracobrachialis corresponding to ossification.
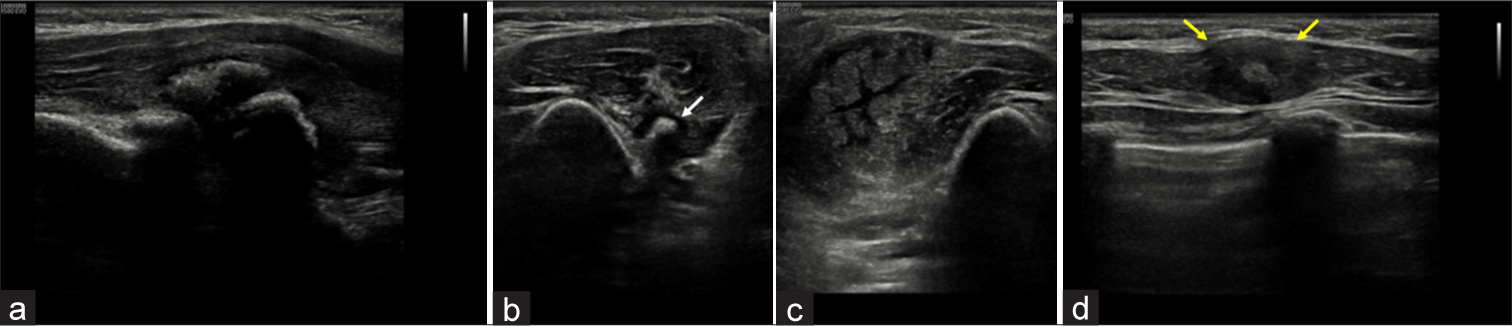
- Ultrasound of arm and chest wall in a 5 year old child with FOP. (a) Grey scale image of the left coracobrachialis muscle showing coarse echogenic areas with dense posterior acoustic shadowing suggesting calcification/ossification. (b,c) Spilt screen image shows (b) osseous lesion in left arm muscles showing ossific focus (white arrows) but no significant edema, (c) comparing it with pre-osseous lesion with extensive edema without ossification in the right arm muscles. (d) Grey scale image of right pectoralis muscle appears focal bulky (yellow arrows) with loss of normal fibrillar pattern and a central echogenic focus, possibly soft calcification.
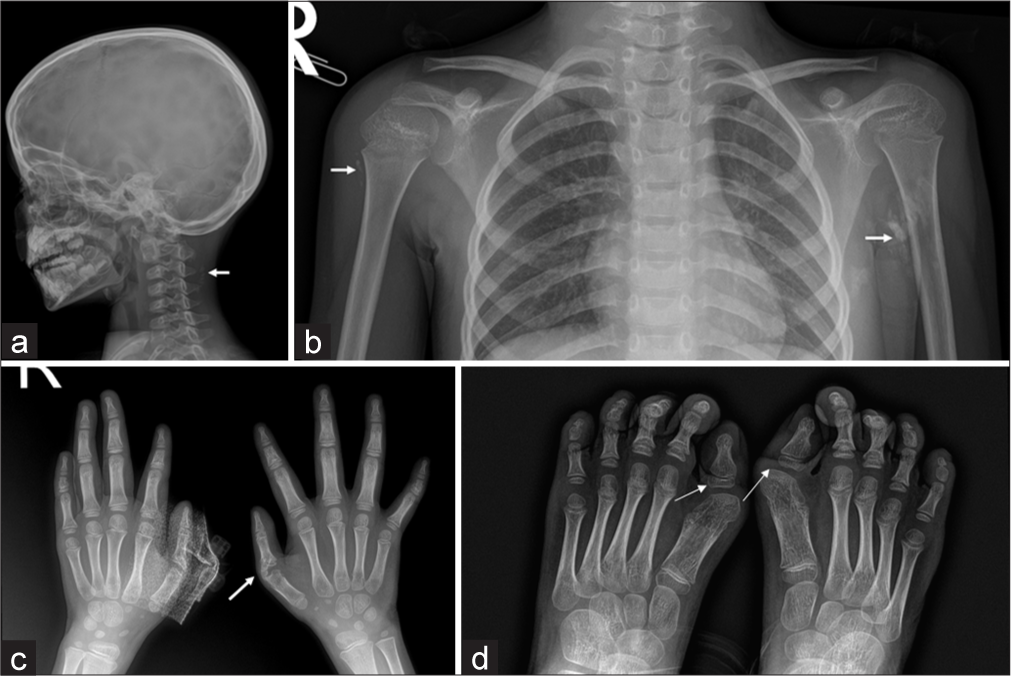
- Radiographs showing skeletal anomalies and soft tissue ossification in a 5 year old child with FOP. (a) Radiograph Skull lateral view, (b) chest with bilateral shoulders shows focal areas of heterotopic ossification (white arrows) in the paravertebral soft tissue and muscles of both upper arms. (c) Radiograph of both hands PA view shows short proximal phalanx of second and fifth digit of both hands with heterotopic ossification (white arrows) bridging across the left first metacarpophalangeal joint. (d) Radiograph of both feet shows short first metatarsals, mono-phalangeal great toe with hallux valgus deformity.
Based on the characteristic skeletal malformations and preosseous as well as osseous soft tissue lesions, the diagnosis of FOP was made and the patient was started on oral steroids and counselled to avoid any trauma and lesion biopsy.
DISCUSSION
FOP is a scare genetic disorder with prevalence of 1 case per 2 million population worldwide.[2,3] It follows an autosomal dominant pattern of inheritance with variable penetrance and most mutations occurring sporadically.[4] Although the pathogenesis largely remains unknown, overproduction of bone morphogenetic protein-4 in lesion cells and lymphocytic cells has been implicated.[5]
Most children present with rapidly growing soft-tissue masses (or flare-ups) in the neck or paraspinal regions between 2 and 5 years of age, which may occasionally settle, but more often progresses to heterotopic ossification, which is usually ascertained radiologically 4–6 months after initial presentation.[4-6] Flare-ups occur spontaneously or are associated with viral illness, post immunizations, intramuscular injections or subsequent to trauma, and fatigue.[1] This eventually leads to progressive immobility, thoracic insufficiency syndrome, recurrent pneumonia, and eventual death, with a median life span being 40 years.[3]
The classical skeletal malformations which are known to be associated with FOP are mono-phalangeal great toes and shortened first metatarsal with hallux valgus (seen in almost all patients with FOP), progressive reduction defects in all digits, webbed toes, shortened thumb, clinodactyly, and shortened femoral neck, cervical spine abnormalities including narrow vertebral bodies, enlarged pedicles, and large spinous processes. These features are usually present at birth, whereas certain abnormalities such as fused posterior elements of cervical vertebrae and tibial osteochondromas may develop later.[1-3,6]
The disease was earlier known as myositis ossificans progressiva which is actually a misnomer as we now know that there is not any muscle inflammation and neither the disease is limited to muscles.[4] In fact, studies have demonstrated that the pre-osseous soft-tissue lesions involve fascia, tendons, ligaments, and skeletal muscle, which may later progress to ossification. The common sites of ossification include sternocleidomastoid, paraspinal, jaw, shoulder, and pelvic girdle muscles and scalp.[3,4] To be more precise, in the skeletal muscles, it is the connective tissue between muscle bundles which is involved, with the changes in muscle being secondary.[4]
MRI appearances depend on the duration of the lesion. The pre-osseous lesions are seen as homogeneous soft-tissue masses of high signal intensity involving the muscles and fascial planes on short-tau inversion recovery and T2-weighted images, which is postulated to be representative of fibroblast proliferation and associated reactive edema. Later on, in the osseous phase, hypointense signals can be seen on T1- and T2-weighted images resulting from fibrous or calcified tissue.[4]
Our patient showed an enhancing soft-tissue mass and multifocal muscle edema as well as multifocal heterotopic ossification, representing pre-osseous and osseous phase of the disease, respectively. There was also a prompt increase in the size of the left chest wall lesion within a week. The radiological appearance had earlier been misinterpreted as infective myositis. However, the characteristic skeletal abnormalities further helped to clinch the diagnosis.
Although MRI signal changes in the soft tissue have been described previously studies, there are only few isolated case reports on the enhancing nature of these pre-osseous masses.[5,7] Pre-osseous enhancing chest wall mass was first reported by Hagiwara et al. in 2003 in a 21-month-old baby with FOP. The enhancement was attributed to the highly vascular fibroproliferative tissue seen in the pre-osseous phase of FOP. The lesion was seen to spread in the subcutaneous region and between muscular bundles predominantly with minimal signal changes in the surrounding muscle fibers.[5]
When encountering such soft-tissue lesions in a child, the differential diagnosis of benign neoplasms such as desmoid tumor and aggressive juvenile fibromatosis as well as malignant masses such as a sarcoma needs to be considered.[4,6] While neoplasms show a gradually progressive growth pattern, preosseous lesions of FOP may show rapid increase or decrease in size, often within hours. MRI imaging features usually point to diagnosis before the appearance of ossification. A diagnostic biopsy should be avoided as it may do more harm to the patient by inciting ossification due to the iatrogenic trauma.[4,5] Moreover, histopathologically, lesions of FOP can be misdiagnosed as desmoid type fibromatosis or sarcoma.[4,6] MRI may help in such cases as the soft-tissue mass of fibromatosis shows poor margination and a heterogeneous signal compared to those of FOP.[4]
Due to the presence of multifocal edema of muscle, another imaging mimic of FOP is idiopathic inflammatory myositis (IIM), including dermatomyositis. Contrary to FOP, in IIM, the pattern of muscle involvement is typically bilateral and symmetric and affects the pelvic girdle and thigh muscles before upper limb and neck muscles with edema of muscles being the predominant MRI abnormality. The fascia and subcutaneous plane may be edematous and commonly have soft-tissue calcifications in patients of dermatomyositis; however, the typical enhancing pre-osseous soft-tissue masses of FOP have not been described.[8] Besides, FOP mainly affects scapular, chest wall, shoulder girdle, and neck muscles in an asymmetric pattern as is well demonstrated in our case.
Clinically and radiologically, rapidly changing soft-tissue masses in the head, neck, or back, along with characteristic skeletal malformations in a child is virtually diagnostic for FOP.[2,4,6] Confirmatory genetic tests are also now available to diagnose FOP in patients with malformed toes before appearance of heterotopic ossification or even soft-tissue flare ups. Genetic sequence analysis found that the children with FOP had the recurrent FOP missense mutation, a single nucleotide substitution (c.617G >A) at codon 206 in the glycineserine activation domain of activin receptor (activin a receptor type 1), a bone morphogenetic protein type 1 receptor.[6]
Awareness of the radiological appearances of FOP is essential as many cases are diagnosed late leading to delayed care. Kitterman et al. have reported that 87% patients of FOP initially received an erroneous diagnosis (which included cancer, fibromatosis, myositis, calcified hematoma, etc.), 67% underwent unnecessary invasive diagnostic procedures, and 54% were subjected to harmful therapeutic interventions such as surgery, chemotherapy, and physical therapy.[2]
Early diagnosis ensures early management, which rests primarily on evasion of soft-tissue trauma, including intramuscular injections, preschool intramuscular immunizations, injections into the jaw for dental work, and unwanted lesion biopsies.[2,4,6] Although there are no established guidelines for medical treatment, radiological improvement has been reported with use of corticosteroids.[4]
CONCLUSION
MRI and sonographic appearances of pre-osseous soft-tissue lesions of FOP may aid in early diagnosis before ossification is visible radiographically, which allows early institution of care to avoid development of debilitating heterotopic ossifications. One should be mindful to avoid misinterpretation of such masses to be of neoplastic etiology and refrain from performing invasive diagnostic procedures.
Ethical approval:
Institutional Review Board approval is not required.
Declaration of patient consent:
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest:
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation:
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship:
Nil.
References
- Imaging assessment of fibrodysplasia ossificans progressiva: Qualitative, quantitative and questionable. Bone. 2018;109:147-52.
- [CrossRef] [PubMed] [Google Scholar]
- Iatrogenic harm caused by diagnostic errors in fibrodysplasia ossificans progressiva. Pediatrics. 2005;116:e654-61.
- [CrossRef] [PubMed] [Google Scholar]
- Fibrodysplasia ossificans progressiva: Initial presentation with a preosseous lesion of the scalp and its MRI appearance. Skeletal Radiol. 2016;45:991-6.
- [CrossRef] [PubMed] [Google Scholar]
- Pre-and post-therapy MR imaging in fibrodysplasia ossificans progressiva. Pediatr Radiol. 2006;36:1108-11.
- [CrossRef] [PubMed] [Google Scholar]
- Contrast-enhanced MRI of an early preosseous lesion of fibrodysplasia ossificans progressiva in a 21-month-old boy. Am J Roentgenol. 2003;181:1145-7.
- [CrossRef] [PubMed] [Google Scholar]
- Early diagnosis of fibrodysplasia ossificans progressiva. Pediatrics. 2008;121:e1295-300.
- [CrossRef] [PubMed] [Google Scholar]
- Teaching NeuroImages: MRI in fibrodysplasia ossificans progressiva. Neurology. 2010;74:e20.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnostic imaging of inflammatory myopathies: New concepts and a radiological approach. Curr Rheumatol Rep. 2019;21:8.
- [CrossRef] [PubMed] [Google Scholar]







