Translate this page into:
MR Findings in the Bone Marrow of a Patient with Anorexia Nervosa

-
Received: ,
Accepted: ,
How to cite this article: Ng CY, Kuek DK, Suresh P. MR Findings in the Bone Marrow of a Patient with Anorexia Nervosa. Indian J Musculoskelet Radiol 2020;2(2):136-9.
Abstract
Gelatinous transformation of bone marrow (GTBM) is a hematological condition found to be associated with states of cachexia and malnourishment, which can be seen in patients with eating disorders, alcoholism, malignancy, and other systemic diseases (such as AIDS, tuberculosis, chronic kidney disease, and chronic heart failure). GTBM is not disease-specific but is a good marker of a severe underlying disease. Initially thought to be a rare finding, newer literature is now demonstrating more cases of GTBM over the past 2 decades, and the alleged rarity may have been attributed to the lack of clinical suspicion and awareness of this condition. We present a case of a young adult female, who has a background of anorexia nervosa and presented with a 4-month history of pain over her left shin. Magnetic resonance imaging (MRI) of the tibia demonstrated the gelatinous transformation of the bone marrow. In this case report, we aim to highlight the underlying pathogenesis of GTBM and its prevalence, its unique distribution within the marrow, its characteristic MRI findings, and how these findings may differ in comparison to normal reconversion marrow and neoplastic infiltration.
Keywords
Bone marrow
Pathology
Gelatinous transformation of the bone marrow
Starvation marrow
Anorexia nervosa
Magnetic resonance imaging
INTRODUCTION
Gelatinous transformation of bone marrow (GTBM), also known as serous atrophy of the bone marrow or starvation marrow, is associated with states of nutritional deprivation and protein catabolism; seen in anorexia nervosa, malignancy, alcoholism, chronic infection (e.g., tuberculosis and AIDS), malabsorption, chronic kidney, and heart disease.[1] It is a good marker for severe underlying disease, although not disease-specific.
Initially described as a rare finding, Shergill et al.[2] demonstrated that in the past 2 decades, more cases of GTBM have been reported, with an increasing incidence from 0.2% to 4.8%.[2] It is suspected that GTBM has been underdiagnosed in the past, which resulted in the alleged rarity. We also suspect with the increasing usage of magnetic resonance imaging (MRI) routinely, we may be detecting more cases of GTBM incidentally, as shown in our case.
This increases the need for the radiologists to be familiar with the characteristic findings of GTBM, which will reduce any unnecessary additional investigations.[3] We aim to highlight these MRI findings and other diagnostic aids described in the literature to help increase the confidence of radiologists in making the diagnosis of GTBM.
CASE REPORT
A female in her 30’s was reviewed in the orthopedic clinic for a 4-month history of left shin and knee joint tenderness. The pain was worse on weight-bearing and mobilizing. Initial plain radiograph of the lower limb demonstrated osteopenia of the bones and loss of fat planes with reduced soft-tissue bulk, but no periosteal reaction. This was followed by an ultrasound scan of the joint which did not identify any cause for her symptoms. The patient had a history of anorexia nervosa, where she was thoroughly investigated for her weight loss over the course of 3 years. She had a BMI of 13.7 and a bone mineral density T score of ‒5.5, in keeping with underlying osteoporosis. The previous blood results demonstrated reduced white and red cell counts and low Vitamin D levels. Given the clinical history, a stress fracture was suspected and an MRI was suggested.
The MRI was performed with axial and sagittal T1w, T2w fat-saturation, and STIR sequences. The T1w sequences demonstrated focal areas of low signal within the marrow of the left tibial diaphysis [Figure 1a]. Corresponding T2w fat-saturation and STIR sequences demonstrated these focal areas as a high signal [Figures 1b and 2] and were found to be present in both lower limbs [Figure 2]. Furthermore, the minimal subcutaneous fat [Figure 1] demonstrated intermediate to low T1w signal and showed little fat-suppression [Figure 3 demonstrates normal marrow and fat signals for comparison]. There was no evidence to suggest an underlying fracture or reactive bone sclerosis. Given the clinical history, distribution, and characteristic MRI signal patterns, the corroborative findings were thought to be most in keeping with GTBM.
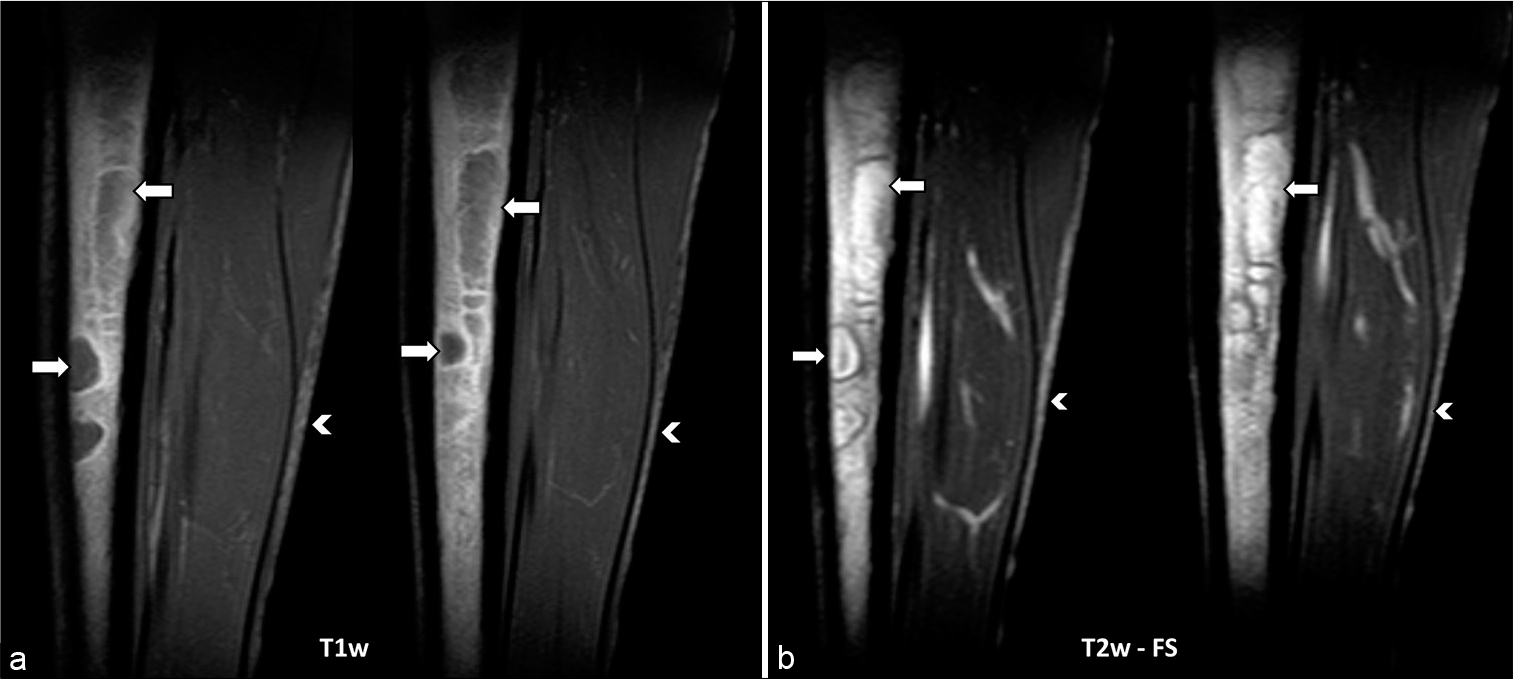
- A young adult female with anorexia nervosa presented initially with pain over her left shin. (a) Sagittal T1w images and (b) fat-suppressed T2w images of the left tibia demonstrate patchy focal lesions predominantly fluid signal intensity (white arrows) within the marrow of the diaphysis. The white arrowheads demonstrate intermediate/low T1w signal in (a) and unsuppressed high signal of the depleted subcutaneous fat in (b).
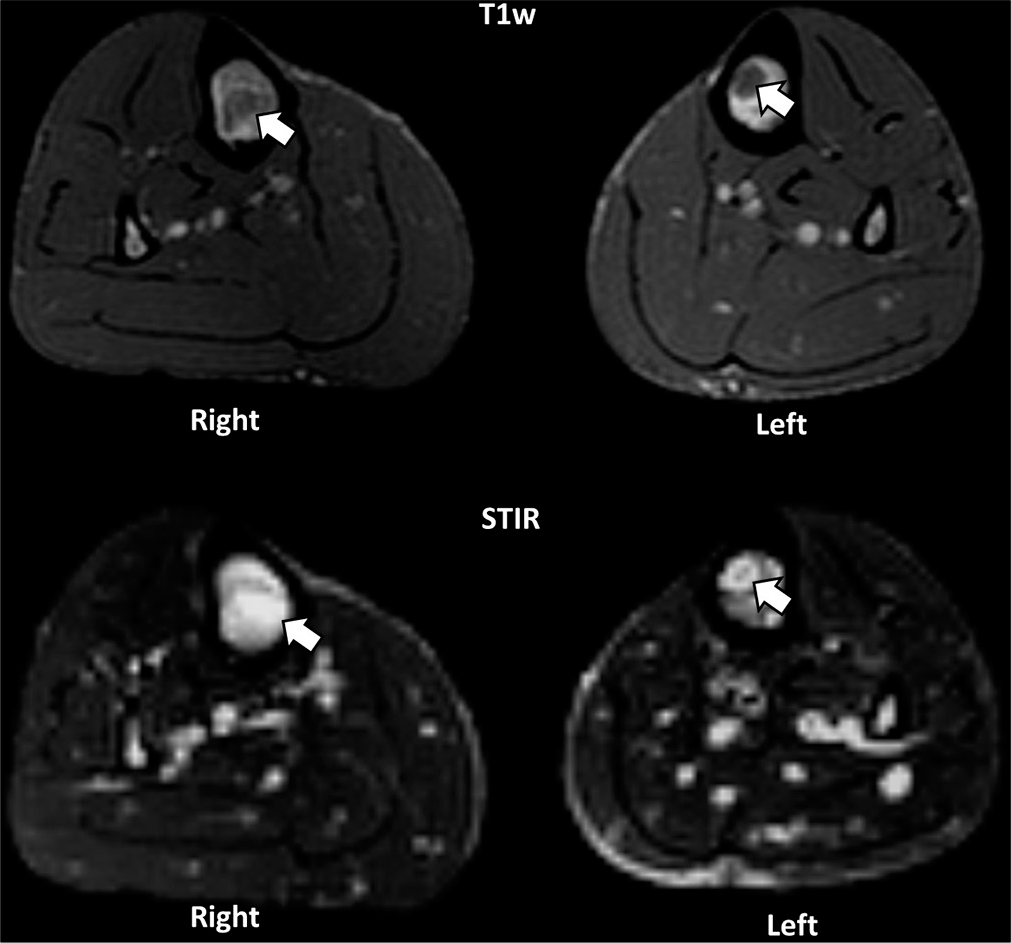
- Axial T1w and STIR images demonstrate abnormal marrow signal (white arrows) in both tibias, confirming a bilateral distribution of the lesions.
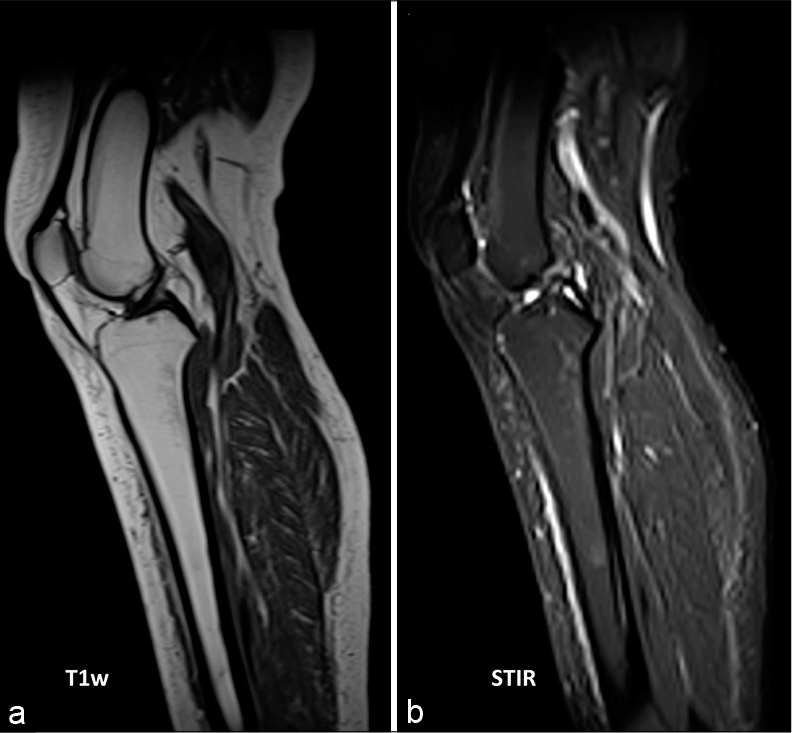
- A healthy 37-year-old female with no significant medical history. (a) T1w and (b) STIR images demonstrate normal fatty marrow which is bright on T1 and dark on STIR and normal subcutaneous fat signal within the lower limb. Please note the signal intensity in contrast to the GTBM changes in anorexia in Figure 1a and b.
DISCUSSION
GTBM has mainly been diagnosed in the past through bone marrow aspirates or biopsies.[2] However, with the increasing usage of MRI and its fat-suppression fluid-sensitive sequences, GTBM may be increasingly detected on MRI.[3-6] Although there is not yet a universally established formal diagnostic criteria for MRI diagnosis, recent literature has consistently described characteristic marrow changes on MRI. At present, bone marrow biopsy remains the gold standard with its specific findings of adipose cell atrophy, hematopoietic tissue hypoplasia, and deposition of extracellular gelatinous matrix.[2]
It was theorized that mobilization of fat stores within the marrow occurs after continuous starvation.[3] This occurs in the late phase of starvation (Phase 3), after fat reserves are depleted and protein catabolism begins. This explains the regression of adipose cells found within the marrow, which is important maintainers of hematopoietic progenitor cells. The concurrent replacement with gelatinous matrix is thought to further inhibit the interaction between remaining hematopoietic cells and signalizing molecules, thus being an adverse microenvironment for hematopoiesis.[1,4,7] Consequently, peripheral blood cytopenias can occur in patients with GTBM, although this is not necessarily a specific or sensitive finding.[8] Our patient had low white and red cell counts but normal platelets.
The study performed by Abella et al.[8] examining 44 patients with anorexia nervosa, demonstrated a range of histological patterns within the marrow of these patients, and suggesting an underlying spectrum: Ranging from normal, bone marrow hypoplasia, and aplasia to GTBM. Of these patients, 89% had abnormal marrow changes, of which 23% were GTBM. Interestingly, the study found that the degree of severity/extensive marrow change was not related to the length or chronicity of disease but the amount of ponderal loss (i.e., the difference between the patients’ actual weight and ideal weight).[8] Studies have shown that GTBM can be reversed after successful treatment and reestablishment of adequate nutritional intake.[1,2,8]
Given the regression of fat and deposition of the gelatinous matrix in GTBM, it is unsurprising that MRI can be useful for detecting these changes. This was first demonstrated by Berg et al.[4] (1995). Characteristically within the abnormal marrow, there are low T1w signal, high T2w signal, and high signal on fat suppression sequences (e.g., STIR and fat-saturation). These changes have a unique distribution and pattern, which are important to not only help diagnose GTBM, but distinguish it from other bone marrow disorders. The study demonstrated that marrow changes affected the distal lower limb bones more than the proximal bones. Furthermore, if there is diffuse involvement of a proximal bone, it was consistently shown that the adjacent distal bones will have diffuse involvement too.[4] This distribution pattern is similar to normal marrow conversion from red to yellow marrow in the first 25 years of life (where conversion starts distally in the appendicular skeleton). This unique characteristic distribution pattern of GTBM differs from physiological marrow reconversion and bone marrow disorders – where their distribution begins typically in the axial skeleton containing remnants of red marrow, whereas GTBM begins in the fatty marrow found in the appendicular skeleton [Table 1].[3-5] Hence, it has been suggested that if diffuse high free water content is observed in the axial skeleton, the diagnoses of underlying GTBM or neoplastic infiltration can be differentiated by obtaining an MRI of the feet – where GTBM is more likely if similar signals are present in the lower limbs.[4] It is important, however to note that there can be either diffuse or focal changes in GTBM depending on the extent of marrow change. In contrast with diffuse involvement, it was found that patchy focal marrow transformation, which most commonly affects the talus and tibia, do not necessarily have associated distal bone changes.[4] Furthermore, if GTBM is found within the long bones, they tend to start in the diaphysis.[3-5] GTBM is also almost always bilateral and symmetrical in the lower limbs (making other pathologies such as infection, tumor, infarcts, or fractures unlikely).[4] There is no contrast enhancement in GTBM and no associated soft-tissue mass or cortical erosion.[3,5]
| Diagnosis | T1w | STIR or T2 with FS | Distribution |
|---|---|---|---|
| Normal marrow conversion (first 25 years of life) | Diffuse high signal (fatty marrow) | Low signal (suppressed fatty marrow) | Starts distally in the appendicular skeleton |
| Normal marrow reconversion (smokers, anemia, etc.) | Diffuse low signal (red marrow) | Intermediate signal intensity | Starts typically in the axial skeleton |
| GTBM (nutritional deprivation and protein catabolism) | Focal or diffuse low signal (gelatinous matrix) | Focal or diffuse high signal | Starts in the fatty marrow found in the appendicular skeleton |
| Lymphoproliferative neoplasm (lymphoma, leukemia etc.) | Focal or diffuse low signal (tumor infiltrate) | Focal or diffuse high signal | Starts typically in the axial skeleton |
With regard to extra-skeletal findings, we see concurrent diminished subcutaneous fat (e.g., Kager’s fat pad), and the fat itself often exhibits signal intensities similar to that of GTBM.[5,6]
Misinterpretations of GTBM as technical failure secondary to failed fat suppression have occurred in some published cases.[2,3] A method recommended by Cheong and Chong[5] to avoid this diagnostic pitfall is to use two separate fat-suppression techniques, which includes a STIR due to its reduced susceptibility to artifact.[5]
GTBM is often shown to be associated with increased risk of stress fractures due to underlying diseases such as anorexia nervosa and how bone marrow composition itself can affect the biomechanical strength of the bone.[3,6] We think that the increased use of routine MRI for suspected stress fractures, especially in anorexia nervosa, is where radiologists would now more commonly pick up incidental cases of GTBM, such as in our case. The abnormal GTBM signals in MRI make diagnosing stress fractures challenging, as the similarly low T1w and high STIR signal seen in marrow edema can be obscured.[3,6] In Bernhard and Cassar-Pullicino’s[6] case, an underlying stress fracture can only be confidently diagnosed if there was reactive bone sclerosis.[6] In the absence of bone sclerosis, which usually takes several weeks to develop, fractures are more difficult to identify. Various authors in such cases have recommended a bone scan or CT if the diagnosis remains uncertain.[3,6] Furthermore, not all patients in this population with limb pain will have detectable fractures.[3] Although it is unclear whether GTBM itself is a direct cause of musculoskeletal pain, osteoporotic pain can certainly occur secondary to undetectable microfractures, muscle fatigue, or hyperalgesia due to the osteoporotic state itself.[9]
CONCLUSION
According to newer literature, GTBM may be under-recognized. With the increasing use of MRI, we may be identifying more cases of GTBM incidentally, which can be a first presentation of an undiagnosed underlying disease, such as anorexia nervosa. Understanding the characteristic MRI findings and unique distribution of GTBM will greatly increase the confidence of the radiologists in making a diagnosis. Furthermore, where there is underlying GTBM, caution should be taken when assessing for stress fractures commonly seen in anorexia nervosa.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Gelatinous transformation of bone marrow. Autops Case Rep. 2017;7:5-8.
- [CrossRef] [PubMed] [Google Scholar]
- Gelatinous transformation of bone marrow: Rare or underdiagnosed? Autops Case Rep. 2017;7:8-17.
- [CrossRef] [PubMed] [Google Scholar]
- MRI findings of serous atrophy of bone marrow and associated complications. Eur Radiol. 2015;25:2771-8.
- [CrossRef] [PubMed] [Google Scholar]
- Distribution of serous like bone marrow changes in the lower limbs of patients with anorexia nervosa: Predominant involvement of the distal extremities. AJR Am J Roentgenol. 1996;166:621-5.
- [CrossRef] [PubMed] [Google Scholar]
- Serous Atrophy of Bone Marrow: A Rare Disorder Worth Getting Acquainted With. Switzerland: European Congress of Radiology EPOS; 2017. p. :2535.
- [Google Scholar]
- Marrow changes in anorexia nervosa masking the presence of stress fractures on MR imaging. Skeletal Radiol. 2006;35:857-60.
- [CrossRef] [PubMed] [Google Scholar]
- Prominent gelatinous bone marrow transformation presenting prior to myelodysplastic syndrome: A case report with review of the literature. Int J Clin Exp Pathol. 2013;6:1677-82.
- [Google Scholar]
- Bone marrow changes in anorexia nervosa are correlated with the amount of weight loss and not with other clinical findings. Am J Clin Pathol. 2002;118:582-8.
- [CrossRef] [PubMed] [Google Scholar]
- Pathomechanisms and management of osteoporotic pain with no traumatic evidence. Spine Surg Relat Res. 2017;1:121-8.
- [CrossRef] [PubMed] [Google Scholar]






